1 Fundamentals of Chemistry
1.2 Molecules
1.3 Measurements
1.5 Periodic Table
1.6 Conversions
1.7 Solutions and their Concentrations
1.10 Balancing Chemical Reactions
1.11 Stoichiometry
1.12 Limiting Reactant
1.14 Chemical Formulas
1.15 Nomenclature
1.42 Learning Outcomes
❮ previous chapter next chapter ❯
external links
Nomenclature
Chemical nomenclature is the naming of Chemical substances. There are tons of resources for you to check out and learn how to name simple compounds and molecules. You can start with the gchem site that has a section on nomenclature. After that, you should check out the OpenStax chemistry book for even more info on how to name compounds. Yes, even Wikipedia has a section on chemical nomenclature although maybe more esoteric.
I do have a nice listing in the Appendix (Chapter 10) of some common polyatomic ions that are used in naming many common ionic compounds. There is also a listing of the some common alkanes. You should memorize these simple sets (ok, just the first 10 alkanes) as a starting point for nomenclature.
Metal Cations with only ONE Oxidation State
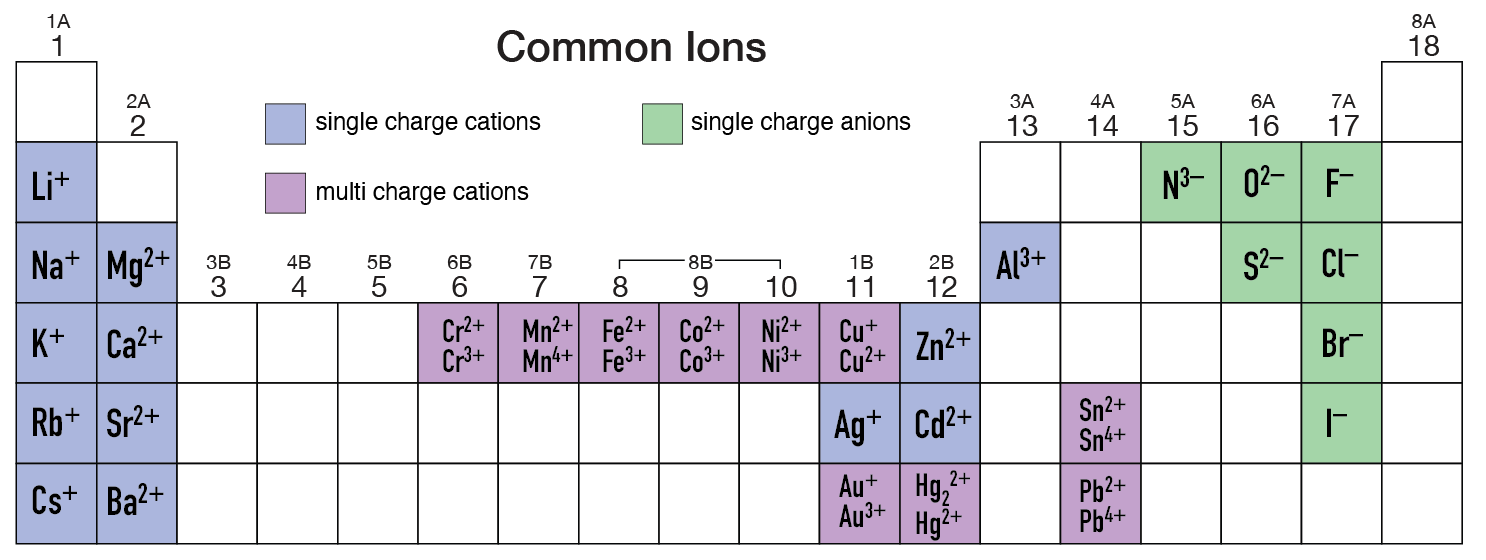
This is important and often overlooked by many folks trying to name an ionic compound. Many metal cations only have one oxidation state (charge in this case) and therefore do NOT have the charge in parenthesis like most of the transition metals. So exactly who are these one-state metal cations. Here you go...
- The Alkali Metal (Group 1) Ions are always +1.
- The Alkaline Earth Metal (Group 2) Ions are always +2.
- A few scattered other metals only have one state and they are...
- Al3+, Zn2+, Ag+, Cd2+
Below is a little periodic table graphic showing the many of the common simple monatomic cations (metals) and anions (nonmetals). All the ions with just one type of charge do not use the parenthesis notation when naming. Example: CdCl2 is called cadmium chloride and not cadmium(II) chloride.